MODERN TRIBOLOGY HANDBOOK Volume Two Materials, Coatings, and Industrial Applications*
- catalytic action of the sliding surface (Zhao et al., 1999a, 2000; Zhao and Bhushan, 2000a, 2001). It is known that a wide variety of materials become electron emitters after mechanical treatment. The term "exoelectron emission" originates from investigation of the emission in freshly formed metals that were produced as a consequence of an exothermal transformation process of the surface. As reported in the literature, exoelectron emission occurs when a material surface is disturbed by plastic deformation, abrasion, fatigue cracking, or phase transformation. The electron emission from freshly formed surface bursts almost immediately during sliding and then decays suddenly as sliding stops. Electron emission has been observed from both metals and non-metals. The energy of exoelectrons is considered to be very low, on the order of 1 to 3 eV.
- Vurens et al. (1992b) studied the decomposition of PFPE lubricants by means of mass spectrometry and FTIR. The gaseous products being generated during sliding (mechanical decomposition) were compared with products from thermal decomposition and from electron decomposition of the PFPE lubricants. They concluded that electron decomposition of perfluoropolyethers gives rise to a different set of products than thermal decomposition. Electron decomposition and thermal decomposition of the lubricants probably occur through different pathways. They suggested that electron decomposition occurs through the formation of a negative ion resistance state, and then a negative ion is formed through dissociative electron attachment and a reactive radical species is generated. Subsequently, gas products are generated from the reactive radical species as well as from the negative ions. The wear fragments suggest that the decomposition of the PFPE lubricants during sliding is an electron-mediated process because most of the product fragment distribution is very similar to the electron decomposition fragment distribution. The electrons necessary for this decomposition could be tribo-electrons generated during the sliding process.
- To study the effect of low-energy electrons, Zhao etal. (2000) and Zhao and Bhushan (2001) conducted sliding experiments in a high-vacuum environmental tribotester and monitored desorbed species with a mass spectrometer. They used a low-energy ultraviolet (UV) lamp to generate low-energy electrons (0 to 5 eV). The illumination of the far-UV light creates low-energy electrons by excitation of the disk surface by UV photons. The produced low-energy electrons then interact with the lubricant molecules and accelerate the decomposition of the lubricant during the sliding process. They reported that UV illumination decreased the durability of the interface; more gaseous fragments were generated; and the partial pressure of the gas fragments was increased because of UV illumination. These results clearly show that UV illumination, which generates low-energy electrons, accelerates the degradation of the lubricant. Thus, triboelectric reaction can be a dominant degradation mechanism. The interaction between the PFPE lubricants and low-energy electrons generated in sliding process is schematically shown in Figure 38.70 (Kajdas and Bhushan, 1999; Zhao and Bhushan, 2001).
- Free radicals can recombine with other radicals, react with PFPE molecules, decompose or interact with low-energy electrons to form other anions.
- Anions react with the positively charged sites of the surface.
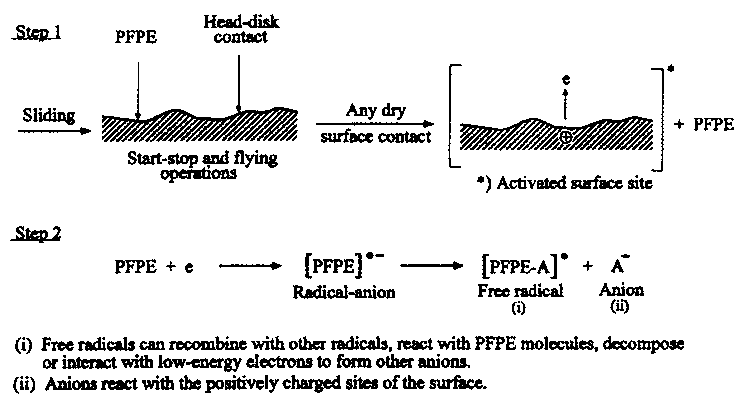
FIGURE 38.70 Schematic showing the electron-induced degradation process of PFPE lubricants.
38.6.4.2.3 Mechanical Scission
Shear rates during start-stops and isolated contacts during flying are high, which can result in mechanical scission. The lubricant film thickness is typically 1 to 2 nm and at 1 to 20 m/s sliding velocity, the shear rate is 5 x 107 to 2 x 109 s -1. At such shear rates, there can be significant energy imparted to the polymer chain, inducing the chains to slide over one another. If the molecules are strongly interacting with the surface, the sliding may be hindered. Additional hindrance to interchain sliding can be the presence of a bulky side group such as the —CF3 group on the Y lubricant. The additional intermolecular friction of the chains with the side group can hinder the rapid configurational adjustments required to support such high shear rates. The chains can be broken by tearing bonds apart along the polymer backbone and reduced to volatile products that provide the route for the observed lubricant loss when contact occurs in sliding or flying. Moreover, potential differences of up to 0.1 V are measured on thin-film disk surfaces, between areas on and off the sliding tracks. Corresponding electric fields for slider/disk separations lead to alteration of organic materials when the localized currents pass between the disk and slider asperities.- Novotny et al. (1994) compared the mass spectra obtained during sliding experiments with those obtained in thermal desorption and found that the spectrum during sliding at 0.5 m/s resembled the thermal spectrum at 300°C rather than that at 90°C. They speculated that the loss mechanism of the lubricants could be of two types: either transient interfacial temperatures were high enough to evaporate lubricant from the surface or polymer scission-produced fragments were small enough to be gaseous. They anticipated that PFPE lubricants were decomposed by the scission of polymer chains driven by several processes, including high mechanical shear rate, high interfacial temperature, and tribocharging.
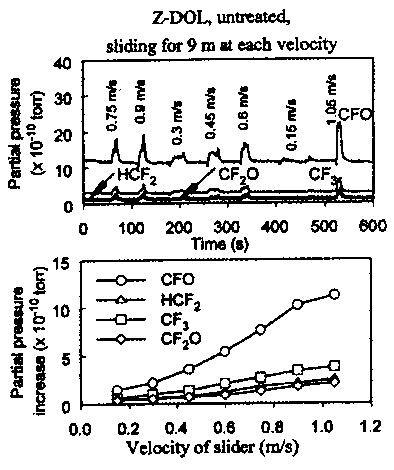
- The effects of mechanical shear were studied by Zhao et al. (2000) and Zhao and Bhushan (2001) by changing the sliding velocity of the slider. The slider slid on different tracks at different sliding velocities, the sliding distance being kept constant at 9 m at different velocities. The test results are shown in Figure 38.71. The partial pressure of mass spectrum peaks in the top figure were produced at different velocities; the sliding velocities are noted at the peaks. The higher the sliding velocity, the stronger the signal peaks. The bottom plot in Figure 38.71 shows the partial pressure increase of four gaseous species as a function of sliding velocity. It is clearly seen that the partial pressure increases with sliding velocity. The partial pressure of the gaseous products is obviously affected by the sliding velocity or mechanical shear rate, and is not affected by the sliding distance or sliding time. These results indicate that mechanical scission is also a dominant degradation mechanism of the PFPE lubricants. A schematic showing PFPE degradation as a result of the mechanical scission is shown in Figure 38.72 (Kajdas and Bhushan, 1999; Zhao and Bhushan, 2001).
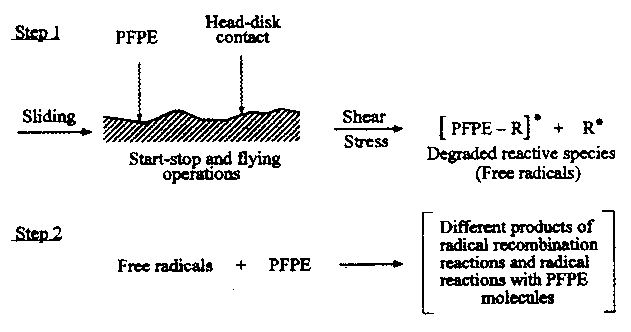
FIGURE 38.72 Schematic showing the degradation process of PFPE lubricants caused by mechanical shear.
38.6.4.2.4 Dominant Degradation Mechanisms
Although several possible mechanisms for decomposition of PFPE lubricants are possible, dominant mechanisms appear to be triboelectric decomposition and mechanical scission. Thermal decomposition may also play a role in contacts at high velocities.38.6.4.2.5 X-1P Additives
X-1P, a cyclic phosphazine lubricant, is commonly used as an additive to PPPE lubricants in thin-film disks. Studies show that lubricant X-1P as an additive increases interface durability, especially at high humidities (Zhao and Bhushan, 1999; Zhao and Bhushan, 2000a). Unlike the long, linear molecular structure of Z-DOL and Demnum lubricants, the X-1P molecule has a dense spherical structure, consisting of six benzene rings and one phosphazine ring. The benzene rings and phosphazine ring are tightly bonded together, forming a whole, firm and dense structure, which makes the X-1P molecule more stable than the Z-DOL molecule. It is difficult for it to be scissioned or to be attacked by low-energy electrons. X-1P's higher viscosity, in addition to its spherical molecule structure, provide for a stronger lubricant film compared to Z-DOL. This minimizes direct solid/solid contact at the interface. - Vurens et al. (1992b) studied the decomposition of PFPE lubricants by means of mass spectrometry and FTIR. The gaseous products being generated during sliding (mechanical decomposition) were compared with products from thermal decomposition and from electron decomposition of the PFPE lubricants. They concluded that electron decomposition of perfluoropolyethers gives rise to a different set of products than thermal decomposition. Electron decomposition and thermal decomposition of the lubricants probably occur through different pathways. They suggested that electron decomposition occurs through the formation of a negative ion resistance state, and then a negative ion is formed through dissociative electron attachment and a reactive radical species is generated. Subsequently, gas products are generated from the reactive radical species as well as from the negative ions. The wear fragments suggest that the decomposition of the PFPE lubricants during sliding is an electron-mediated process because most of the product fragment distribution is very similar to the electron decomposition fragment distribution. The electrons necessary for this decomposition could be tribo-electrons generated during the sliding process.
Editor-in-Chief
Bharat Bhushan, Ph.D., D.Sc. (Hon.)
Department of Mechanical Engineering
The Ohio State University
Columbus, Ohio
Computer Microtribology and Contamination Laboratory, The Ohio State University, 206 W. 18th Avenue, Columbus, Ohio 43210, U.S.A.
CZESLAW KAJDAS
Warsaw University of Technology, Institute of Chemistry in Plock, 17 Lukasiewicza Street, 09-400 Plock and Central Petroleum Laboratory in Warsaw, Poland
* — Published in : B. Bhushan (ed.), MODERN TRIBOLOGY HANDBOOK Volume Two Materials, Coatings, and Industrial Applications, 1489-1491. 2001 CRC Press Boca Raton London New York Washington, D.C. Library of Congress Cataloging-in-Publication Data