Nanoscale Boundary Lubrication of Diamond-like Carbon Coatings with Fluorinated Compounds*
C. Kajdas
Institute of Chemistry and Central Petroleum Laboratory, Warsaw University of Technology, Poland
Abstract
Progress in the technology of magnetic media has brought about a remarkable increase in recording density. The most important factor determining the utility of magnetic disks is durability against head wear, and this durability is controlled by several factors. The present paper discusses the tribology of these media, particularly from the viewpoint of boundary lubrication. In that context there are two characteristic features of this lubrication regime: specific standard lubricants (fluoropolyethers such as Z-DOL and perfluoropolyetliers such as Z-15) and the newer application of these lubricants in the form of films only a few nanometers thick. Advanced phosphazene-type fluorinated compounds are of most interest at present, so these compounds are discussed in more detail. The emphasis is on X-1P lubricant used either alone or as an additive for fluoro- and perfluoropolyethers deposited on protective diamond-like carbon coatings.
Keywords
boundary lubrication, fluorinated lubricants, diamond-like carbon, computer disks
* — Published in : J. Synthetic Lubricants 18-1, April 2001. (18) 18
INTRODUCTION
New sophisticated techniques for measuring friction, wear, lubricant thickness, surface topography, and adhesion (all on a micro- to nanoscale) for imaging lubricant molecules, and the availability of superconductors for atomic scale simulations have led to the development of a new field of research called microtribology, nanotribology, molecular tribology, or atomic-scale tribology [1-3]. The converse of macrotribology, micro/nanotribology studies the wear behaviour of components at least one of which has extremely small mass. Since negligible wear occurs in these circumstances, tribological performance at this scale is mostly controlled by the lubrication of component surfaces. Under these specific nanoscale boundary lubrication conditions, lubricant films are very thin.
Table 1 Chemistry and selected properties of PFPE lubricants Z-15, Z-DOL, and SA, and the X-1P lubricant/additive [6] | |||
| Lubricant | Formula | End groups | Molecular weight |
| Z-15 | CF3—O—(CF2—CF2—O)m—(CF2—O)n—CF3 | —CF3 | 9100 |
| Z-DOL* | HO—CH2—CF2—O—(CF2—CF2—O)m—(CF2—O)n—CF2—CH2—OH (m/n ~ 2/3) | —OH | 2000 |
| SA* | F—(CF2—CF2—CF2—O)m—CF2—CF2—CH2—OH | —OH | 3600 |
| X-1P | 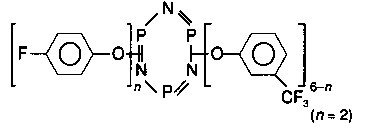 | —CF3 | 1000 |
| Lubricant | Formula | Density (x10³ kg/m³ at 20°C) | Kinematic viscosity (mm²/s) |
| Z-15 | CF3—O—(CF2—CF2—O)m—(CF2—O)n—CF3 | 1.84 | 150 at 20°C |
| Z-DOL* | HO—CH2—CF2—O—(CF2—CF2—O)m—(CF2—O)n—CF2—CH2—OH (m/n ~ 2/3) | 1.81 | 80 at 20°C 34 at 40°C |
| SA* | F—(CF2—CF2—CF2—O)m—CF2—CF2—CH2—OH | 1.87 | 75 at 40°C |
| X-1P |  | 1.48 | 1826 at 20°C 205 at 40°C |
|
| |||
Emphasis is on X-1P lubricant used alone and as an additive for PFPEs deposited on protective DLC coatings. Table 1 shows some PFPE lubricants and the X-1P compound.
PHOSPHAZENE-TYPE COMPOUNDS
Chemistry and general tribological characteristics
Phosphazenes are ring or chain compounds consisting of alternating phosphorus and nitrogen atoms with two substituents attached to phosphorus. Owing to the presence of phosphorus and nitrogen, phosphazenes are inherently fire resistant. Cyclic phosphazenes are either liquids or low melting point crystalline solids. Their physical properties vary considerably with molecular weight and substituents. Phosphazenes are of interest in applications where fire resistance and thermal stability are important considerations. With the proper selection of substituents, thermally and hydrolytically stable ring and chain compounds, including fluids with low pour points and good thermal stability, have been synthesised [7]. Phosphazenes have properties necessary for advanced lubricants and/or additives for high performance applications [7-8]. Recently, a number of substituted aryloxycyclotriphosphazenes were investigated with a view to meeting the lubrication requirements of the integrated high performance turbine engine technology initiative [9]. Consideration of various properties and economic factors led to the identification of a derivative containing four meta-CF3, and two para-F substituents, as the leading fluid candidate.X-1P boundary film formation
Recently, the dynamics of the formation and loss of boundary films formed during the lubricated sliding of steel surfaces were investigated over a range of temperatures and applied loads [10]: X-1P lubricant and mineral oil with and without zinc dialkyldithiophosphate (ZnDDP) additive were used in tests on a
 |
|
|
SELECTED PHOSPHAZENE-TYPE COMPOUNDS AS ADVANCED TOPICAL LUBRICANT ADDITIVES AND/OR LUBRICANTS
General information
Mechanical interactions between the head and the medium are minimised by lubrication of the magnetic medium. Magnetic storage devices used for information (audio, video, and data processing) storage and retrieval are tapes, floppy disks, and hard disk drives. Magnetic media fall into two categories [14]: particulate media, where magnetic particles are dispersed in a polymeric matrix and coated on to a polymer substrate for flexible media (tapes and floppy disks), or on to a rigid substrate (typically aluminium, and more recently glass) for rigid disks; and thin-film media, where continuous films of magnetic materials are deposited on to the substrate by vacuum techniques.X-1P additive and/or lubricant
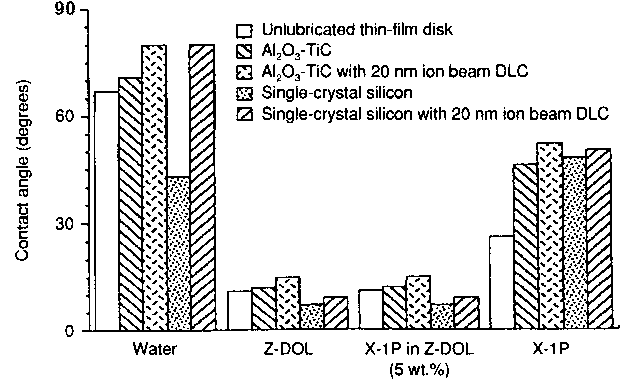
General characteristics The X-1P product is a mixture of p-fluorophenoxy- and m-trifluoromethyl-phenoxy-substituted cyclic phosphazenes. The phosphazene ring in X-1P may exist in a puckered or planar form [25,26]. In the planar form, the phosphazene ring is sterically shielded by the six phenoxy groups, and it is unlikely that strong adhesive interaction occurs between partially fluorinated phenyl rings and the carbon coating. In the puckered form, a basal plane is defined by the nitrogen atoms and three phenoxy groups in the equatorial positions, and the three phenoxy groups in the axial positions are directed upward. A strong interaction is then possible between the exposed phosphazene ring and the carbon coating. Selected properties of X-1P and Z-DOL are presented in Table 1. Advanced analytical techniques have been used to investigate the degradation mechanism and oxidative stability of X-1P cyclophosphazene lubricant [27].Trihological performance A cyclic phosphazene lubricant, X-1P, is considered as one of the most advanced lubricants for both rigid and flexible thinfilm magnetic media. It is a very special non-polymer compound, which can be used either as an additive for regular PFPE lubricant or just as a superb lubricant. Early information on the tribological properties of X-1P was published in 1992 [9]. The first papers considering the unique tribological performance relevant to the disk-drive industry were published in 1994 and 1995 [15-16]. These papers generated significant interest in the application of this special non-polymeric chemical as a lubricant for advanced recording media. It was shown that X-1P by itself performs well on hard disks during CSS and stiction tests with a lubricant thickness of about 0.5 nm [16]. Most of the early work testing X-1P involved its use as a pure lubricant coated by dipping or draining, resulting in a thickness of less than 1 nm. This work was published for both hot/wet environments [15] and ambient conditions [16]. Laptop computing had increased the importance of lubricant performance in adverse environments, especially high humidity (80% RH) and somewhat elevated temperature (30°C). Contact start-stop (CSS) test results with X-1P show it to have a tribological performance similar to or better than Z-DOL, as a topical lubricant on carbon coated thin-film rigid disks under ambient conditions; however, X-1P significantly outperforms Z-DOL in CSS testing under hot/wet (30°C, 80% RH) conditions [15]. Additionally, X-1P was found to be stable when heated in the presence of either water or a standard slider material Al2O3-TiC.
Research emphasises that X-1P is advantageous for use in pseudo-contact recording due to protection of the head [19]. When working with very thin layers the lubricant serves as a boundary layer. It was also stressed that X-1P serves to protect the head from the catalytic decomposition of PFPE-type lubricants.
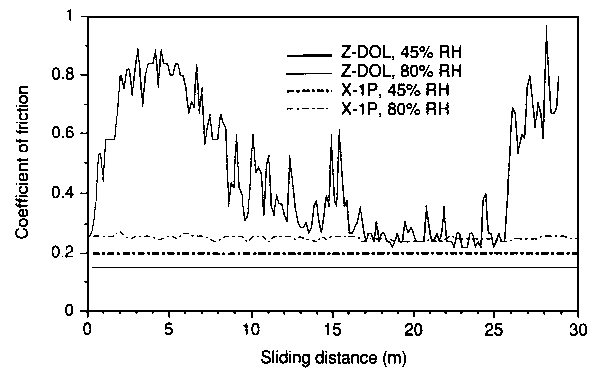 | Figure 3 Ball-on-flat sliding results of Al2O3-TiC wafer against sapphire ball for Z-DOL and X-1P at 45 and 80% RH [22] |
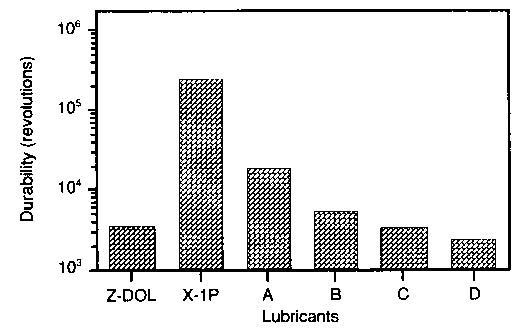
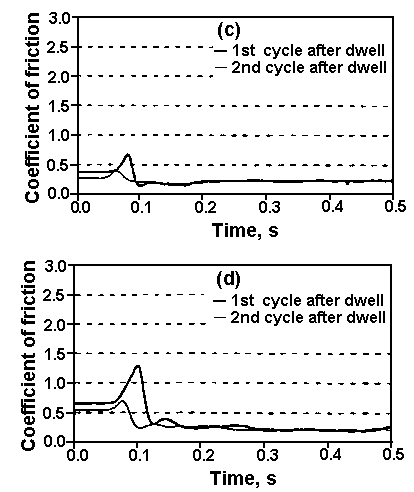
It was found that an X-1P film of 1 nm exhibits a low coefficient of kinetic friction ranging from an initial value of 0.18 to 0.36 at 2 x 105 disk revolutions. The coefficient of friction for Z-DOL, at the same thickness, is initially similar to X-1P but then increases rapidly to reach 0.7 at 7 x 10³ disk revolutions. The coefficient of friction of Z-DOL with X-1P as an additive is initially at the same value; however, it increases rapidly after 104 revolutions to reach 0.55 after 2 x 104 revolutions. Figure 2 shows that X-1P has the highest durability, followed by Z-DOL with 10 wt.% X-1P additive.
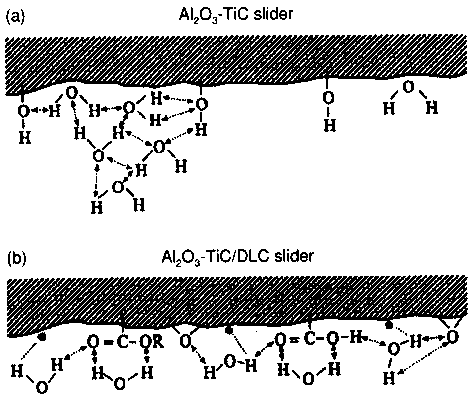
Action mechanism The experimental findings described above have been accounted for by a new mechanism approach with the concept of hydrogen bonding interaction and triboemission [6]. The new mechanism was specially developed for the Al2O3-TiC surface sliding against a disk with Z-DOL lubrication at high environmental humidity. The mechanism, a speculative one, also takes into account the role of X-lP lubricant in tribological performance.
 |
|
-
This mechanism is as follows:
- at the interphase, there exist hydrogen atoms with partial positive charge;
- hydrogen bonding interactions at the sliding interphase results in high friction which depletes the lubricant at some sites;
- low-energy electrons are emitted from the sites with solid-solid asperity contact, including C-O bond scission through the interaction of low-energy electrons with lubricated molecules.
The electron-induced degradation mechanism is based on the mechanism of lubricant component molecules by low-energy electrons [28]. The principal thesis of the model is that the lubricant components, for example, alcohols [29], form anions, which are then chemisorbed on the positively charged areas of rubbing surfaces. The general model of the negative ion-radical action mechanism of lubricating oil components assumes creation of two types of activated sites on the friction surfaces, that is, thermally activated sites and sites activated by the low-energy electron emission process (exoelectrons).
-
Figure 5 Diagram of
- Al2O3-TiC and
- Al2O3-Ti-C/DLC and
(a:C-Hx) disk/Z-DOL interaction via hydrogen bonding [6]
It is assumed that the exoelectron energy is sufficient to cause the ionisation of lubricant molecules.
- Figure 4(a) shows the interaction of water molecules with the Al2O3-TiC surface. Hydroxyl groups are chemisorbed from the environment on to the A12O3-TiC surface. In humid environmental conditions, an absorbed water film interacts with the chemisorbed hydroxyl groups through partially negatively charged oxygen atoms and partially positively charged hydrogen atoms. The higher the humidity, the more interacted water molecules are present.
Figure 4(b) demonstrates that in the case of the A12O3-TiC surface with a DLC coating, groups other than hydroxyl groups (e.g., ester and carboxylic groups) are the most chemisorbed surface species. The water molecules interact by hydrogen bonding with these functional groups, forming a more strongly absorbed film. The water film is additionally strengthened by the interaction of water molecules with dangling bonds in the carbon coating.- The hydroxyl and oxygen groups favour water adsorption so they belong to the hydrophilic category. Z-DOL has hydroxyl groups at both ends while DEMNUM-SA has only one hydroxyl end-group. Additionally, the backbone of Z-DOL has a higher frequency of oxygen linkage in the ether group (number of oxygen atoms per CF2 groups) than DEMNUM-SA. Accordingly, it is expected that Z-DOL will interact more with water molecules. The results of static friction, kinetic friction, and durability tests have confirmed that DEMNUM-SA is relatively insensitive to humid environments, compared with Z-DOL [30]. On the other hand, X- IP has no hydrophilic end groups, so there is no interaction between its end groups and water molecules.
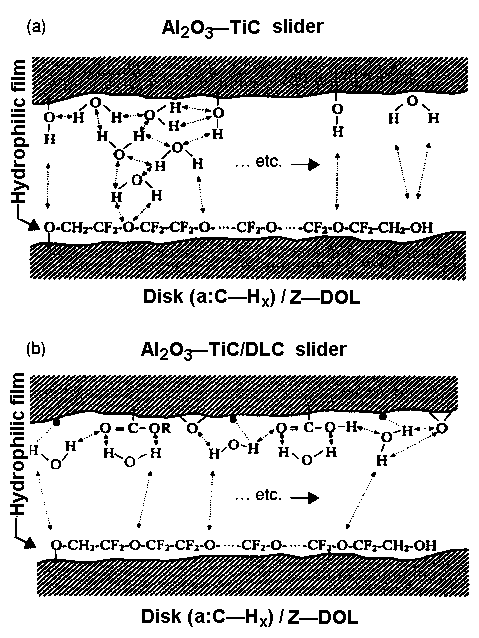
- A diagram of head/disk interaction through hydrogen bonding is shown in Figure 5(a). The head is made of Al2O3-TiC, and the disk is coated with amorphous hydrogenated carbon and lubricated with Z-DOL. On the disk surface there are some functional groups (Figure 6) that also interact with the Z-DOL molecules. The strength of the head/disk interaction is controlled by the water film thickness on the Al2O3-TiC surface: the higher the humidity, the thicker is the water film. Some micro-water-droplets can penetrate the lubricant film, displacing it from the surface. The upward side of Z-DOL film, particularly at the -CF2—CH2—OH end, will interact with the Al2O3-TiC layer.
- Figure 5(b) shows that for an Al2O3-TiC head with carbon coating, the water molecules interact strongly with the functional groups of the carbon coating and dangling bonds (see Figure 6) of the head surface;
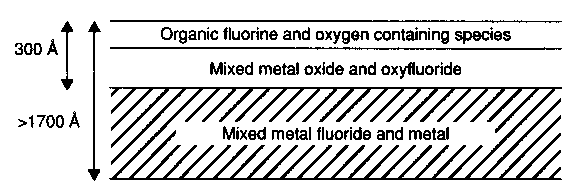
Figure 6 Simplified diagram of the chemical structure of hydrogenated carbon overcoat surface [5] 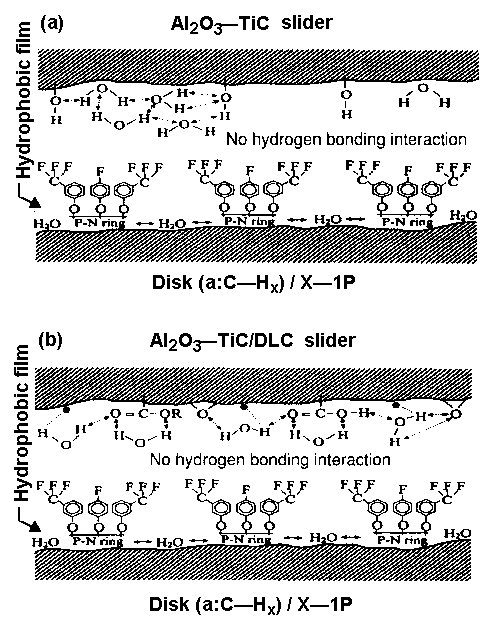
-
Figure 7 Schematic of
- Al2O3-TiC and
- Al2O3-TiC/DLC and
(a: C—Hx) disk/X-lP interaction via hydrogen bonding [6]
as a consequence the hydrogen bonding interaction between the head and disk is significantly reduced compared with Figure 5(a). Less hydrogen bonding prevents high friction at the interface. The lubricant film covers the surfaces and an increase in the electron emission during sliding through solid-solid asperity contact is alleviated. Lack of low-energy electrons postpones the degradation of Z-DOL lubricant molecules.
- Figure 7(a) shows that when X-1P is applied to the disk surface instead of Z-DOL, there is no hydrogen bonding between the two surfaces.
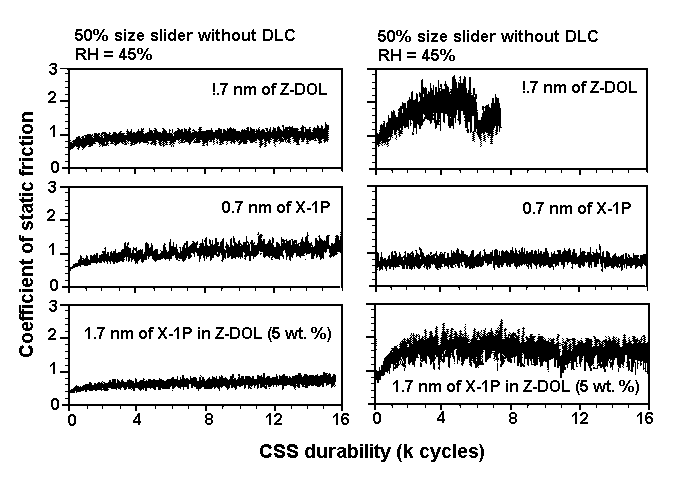
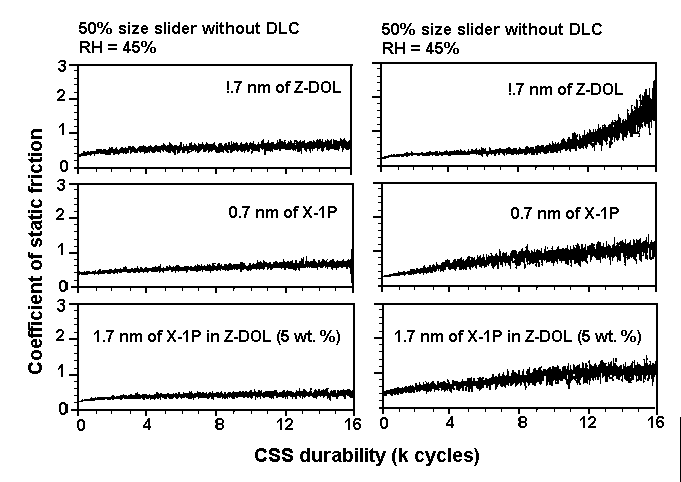
Hydrophobic —CF3 and —F layers repel the water film. Similarly, in the case of an Al2O3-TiC/DLC head and DLC/X-1P disk system ( Figure 7(b)), water molecules are confined to the head slider because no hydrogen bonding occurs at the interface. The role of carbon in confining water molecules is not so critical to tribological performance as for Z-DOL. This accounts for similar tribological performance obtained from CSS tests between an Al2O3-TiC slider without and with the DLC coating at high humidity [22]. CSS test results obtained at 45% and 80% RH, using Z-DOL, X-1P, and X-1P as an additive in Z-DOL, and an Al2O3-TiC slider without and with DLC coating, are depicted in Figures 8 and 9. The mechanism is not substantiated yet, and should be considered speculative. However, as discussed in detail elsewhere [6], it explains the degradation process of Z-DOL, and the lesser degradation of X-1P.
X-100 and XML-86 phosphazene type additives
General information It has been demonstrated above that X-1P shows a lower stiction value and a smaller increase in the coefficient of stiction as a function of CSS cycles than the typical PFPE lubricant Z-DOL. It was also shown that X-1P used as an additive in Z-DOL reduces stiction and increases the stability of Z-DOL, particularly at higher environmental humidity. However, one possible concern with X-1P as an additive in Z-DOL is phase separation, because X-1P is immiscible with Z-DOL. Phase separation of phosphazene additives in PFPE lubrication was investigated as a function of additive concentration and lubricant layer thickness [24]. Two additives, X-1P and X-100, were tested. The amount of phase separation was found to be higher for X-1P than X-100. It was concluded, however, that phase separation can be reduced and possibly avoided if additives and disk properties are chosen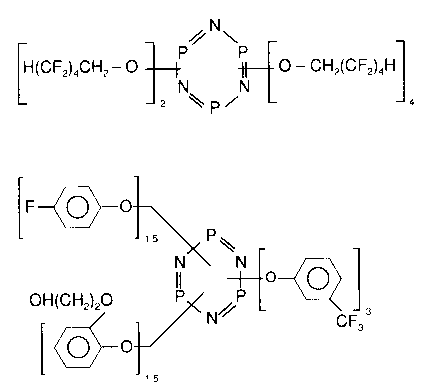
Figure 10 Chemistry of X-100 - (a) and XML-86
- (b) additive compounds [23]
judiciously. This is related to previous results [31] and attributed to the interaction between phosphazenes and the active carbon surface. It was shown that a complex is formed from this interaction, which is stable if the carbon is active when coated; near edge X-ray absorption fine structure analysis was used to investigate the presence of unsaturated carbon bonds at interphases lubricated with Z-DOL and Z-DOL/additives mixtures [24].
Chemistry of X-100 and XML-86 phosphazenes The chemical structures of the X-100 and XML-86 compounds are shown in Figure 10. These are newly developed phosphazene-type lubricants with low vapour pressure and high thermal stability [23]. They have some structural similarities to Z-DOL and therefore better solubility in Z-DOL than X-1P. Comparing Table 1 and Figure 10, it can be seen that X-100 and XML-86 have a phosphazene ring structure that is similar to X-1P, the difference is in the chemistry of the phosphazene ring substituents. X-100 has fluoropolymer substituted side chains [H(CF2)4—CH2—O—] while XML-86 has a branch that is similar to the functional end group of Z-DOL [(HO—CH2—CH2—O— ) group in XML-86 and (HO—CH2—CF2—O—) group in Z–DOL]. These compounds are more soluble in Z-DOL than is the X-1P additive: about 2 wt.% solubility for X-100 and 0.05 wt.% for X-1P in Z-DOL.