Boundary Lubrication of Low-Sulphur
Diesel Fuel in the Presence of Fatty Acids
Czeslaw Kajdas and Marzena Majzner Warsaw University of Technology, Institute of Chemistry, Plock, Poland
Abstract
This paper presents an overview of the effect of aliphatic acids on the tribological properties of selected hydrocarbons and petroleum fractions. The major experimental part of the work focuses on improvements to the lubricity of low-sulphur diesel fuel. Experiments were carried out using a pin-on-disc friction machine and HFRR test rig. The wear results obtained clearly show a specific effect of the test acids dissolved in hydrocarbons in the concentration range 0.005-0.1% (50-1000 ppm). Although the overall picture presented by these results is very complex, it can be concluded that a very small amount of the selected acids dramatically improves low-sulphur diesel fuel lubricity.Keywords: lubricity, diesel fuel, boundary lubrication, aliphatic acids, HFRR, concentrations
Published in : Lubrication Science 14-1 p. 84-107
INTRODUCTION
Boundary lubrication plays a key role in lubrication of diesel fuels. However, there is considerable evidence that viscosity still has some influence, because many pump contacts operate in the mixed lubrication regime, where the hydrodynamic film formed is comparable to the surface roughness. Unfortunately, fuels are inherently poor lubricants from both the hydrodynamic and boundary lubrication point of view [1].| Table 1. Properties of low-sulphur diesel fuel components | ||
| Property | HON component | HK component |
| Density at 20°C, g/cm3 | 0.813 | 0.835 |
| Kinematic viscosity at 40°C, mm2/s | 1.7 | 3.0 |
| Sulphur content, ppm | 76 | 1 |
| CFPP,°C | <-30 | -18 |
| Cetane index | 51 | 52 |
Distillation
T10,°C T50,°C T90, °C | 173 | 198 |
| Table 2. Characteristics of distillation fractions of HON component | ||||
| Fraction | IBP, °C | FBP, °C | at 40°C, mm2/s | Sulphur content ppm |
| I | 173 | 201 | 1.1 | 46 |
| II | 201 | 216 | 1.2 | 58 |
| III | 216 | 221 | 1.3 | 46 |
| IV | 221 | 227 | 1.4 | 46 |
| V | 227 | 238 | 1.5 | 50 |
| VI | 238 | 242 | 1.7 | 56 |
| VI! | 242 | 256 | 1.8 | 44 |
| VIII | 256 | 270 | 2.1 | 78 |
| IX | 270 | 287 | 2.4 | 99 |
| X | 287 | - | 3.3 | 153 |
AIM OF RESEARCH
The objectives of this paper are:- to present the tribological properties of fractions used to produce lowsulphur diesel fuels and to determine the influence of fatty acids on the tribological properties of these components
- to describe the influence of fatty acids on the wear of systems lubricated with fractions obtained by fractional separation of components
- to demonstrate how the chemical structure and viscosity of base fluids influence the antiwear and pro-wear behaviour of fatty acids.
Table 3. Kinematic viscosity of base fluids Base fluid Kinematic viscosity at 40°C, mm2/s 1-Methylnaphthalene 2.1 Mixture of n-hexadecane and 1 -methylnaphthalene (volume ratio 1:1) 2.2 n-Hexadecane 2.9 PA04 16.6 PAO 6 28.9 Saturated hydrocarbon mixture from SN 400 50.4 Mineral base oil SN 400 76.7 EXPERIMENTAL
Lubricant materials
Base fluids Two major components of low-sulphur diesel fuel were tested: the HON component and the HK component. The HON component is the low-sulphur fraction from diesel fuel desulphurisation. The HK component is the ultralow-sulphur fraction from hydrocracking. The properties of low-sulphur diesel fuel components are given in Table 1.- Additionally, tribological tests were carried out for distillation fractions obtained by fractional separation of the HON component (properties of fraction I-X are summarised in Table 2).
- Seven other base lubricants with a wide range of viscosities (Table 3) were used to find out the influence of fatty acids on the wear behaviour of some model saturated and aromatic hydrocarbons along with the typical mineral base oil. These fluids include:
- two pure model hydrocarbons: n-hexadecane and 1 -methylnaphthalene (used as the reference paraffin and aromatic base fluid)
- mixture of n-hexadecane and 1-methylnaphthalene (a low-viscosity hydrocarbon reference base fluid)
- two synthetic hydrocarbon base oils: PAO 4 and PAO 6
- saturated hydrocarbons separated from SN 400 base oil
- mineral base oil SN 400.
Table 4. Characteristics of fatty acids Fatty acid Chemical structure Molecular weight Purity, % Caproic acid CH3-(CH2)4-COOH 116.16 99 Capric acid CH3-(CH2)8-COOH 172.27 ~99 Lauric acid CH3-(CH2)10-COOH 200.32 >99 Palmitic acid CH3-(CH2)14-COOH 256.43 >97 Stearic acid CH3-(CH2)16-COOH 284.49 ≥90 Behenic acid CH3-(CH2)18-COOH 340.61 - Table 5. Solutions of fatty acids in different base fluids investigated Base fluid Fatty acid Fatty acid concentration, ppmt HON component Caproic acid 50, 100, 500, 750, 1000 Capric acid 50, 100, 500, 750, 1000 Lauric acid 50, 100, 500, 750, 1000 Palmitic acid 50, 100, 500, 750, 1000 Stearic acid 50, 100,500,750, 1000 Behenic acid 500 HK component Stearic acid 100, 500, 1000 Fraction I-X of HON component Stearic acid 500 1 -Methylnaphthalene Palmitic acid 50, 100, 500, 750, 1000 Mixture of n-hexadecane and 1-methylnaphthalene (volume ratio 1:1) Palmitic acid 50, 100,500, 750, 1000 n-Hexadecane Palmitic acid 50, 100, 500, 750, 1000 PA04 Palmitic acid 50, 100 ,500, 750, 1000 PA06 Palmitic acid 50, 100, 500, 750, 1000 Saturated hydrocarbon mixture from SN 400 Palmitic acid 50, 100, 500, 750, 1000 Mineral base oil SN 400 Palmitic acid 50, 100,500,750, 1000 Fatty acid solutions Table 4 presents the aliphatic fatty acids used as additives in the present work and the molecular weight and purity of these substances. All the compounds were applied as received, without further purification. Fatty acids were added to base fluids in the concentration range 50-1000) ppm. The solutions of fatty acids tested are listed in Table 5.
Table 6. Pin-on-disc experimental set-up and test conditions Material system 52100 steel-on-52100 steel Geometry Sphere-on-flat (fixed ball on rotating disc) Specimens - ball
- disc
Diameter: 3.18 mm; Ra = 0.3-0.35 µm; hardness: 63 HRC
Diameter: 25.4 mm; Ra = 0.2 µm; hardness: 58-63 HRCWear track radius, mm 8 Applied load, N 9.81 Sliding velocity, m/s 0.25 Sliding distance, m 500 Temperature, °C 25 Apparatus and test conditions
The wear tests were carried out using:- HFR2 apparatus (made by PCS Instruments, UK)
- pin-on-disc contact machine (Tester T-01 M made at the Terotechnology Institute, Radom, Poland).
- The test conditions were chosen according to CEC F-06-A-96 'Measurement of diesel fuel lubricity - approved test method. HFRR fuel lubricity test'.
Pin-on-disc contact machine The specimens (a stationary ball and a rotating disc) were clamped in place with holders. The disc holder contained the test material, fully flooding the contact region. A dead-weight loading system applied normal force. The sliding velocity and temperature (programmed temperature 25°C) were recorded continuously during the test.
- The test conditions (summarised in Table 6) were designed to result in boundary lubrication at the sliding interface.
Procedure
Prior to use, the test specimens were ultrasonically cleaned in toluene for 20 min and in acetone for 20 min. The same cleaning procedure was applied to the holders before testing each lubricant. Tribological tests using the HFR2 apparatus were carried out under test conditions in accordance with CEC F-06-A-96: 17.0°C < temperature < 23.5°C; absolute humidity > 0.8 kPa, relative humidity <: 70%; 23.5°C < tem- perature ^ 28.0°C; and 0.8 kPa < absolute humidity < 2.0 kPa. The same con- ditions were selected for tests with the pin-on-disc contact machine. A minimum of two tests was done using the HFR2 apparatus and a min- imum of five tests was carried out by the pin-on-disc contact machine for a test material.Interpretation
HFR2 apparatus test outputs The wear-scar diameter was measured in both the X and the Y directions using a photomicroscope to an accuracy of ±1 µm, and the mean wear-scar diameter (MWSD) was evaluated:MWSD =(X + Y)/2 (1) In accordance with CEC F-06-A-96, the corrected wear-scar diameter was calculated in um as follows:
WS1.4 = MWSD + HFC x (1.4 -AVR) (2) where HFC is the humidity correction factor in µm/kPa (for unknown fuel samples, ?HCF = 60 µm/kPa); and AVP is the mean vapour pressure during the test in kPa, given by
AVP = (ACT1 + AVP2)/2 (3) AVP1 is the absolute vapour pressure at the start of the test in kPa, given by
AVP2 = (%RH2 x 10n)/750 (4) where %RH1, is the relative humidity at the start of the test, and n is given by
n = 8.017352- [1705.984/(231.8636 + T1)] (5) where T1, is the air temperature at the start of the test in °C. AVP2 is the absolute vapour pressure at the end of the test in kPa, given by
AVP2 = (%RH2 x 10 n )/750 (6) where %RH2> is the relative humidity at the end of the test, and
v = 8.017352 - [1705.984/(231.8636 + T2)] (7) where T2 is the air temperature at the end of the test in °C.
- The volume of worn material from the ball (volume wear, VW) was calculated from the corrected wear-scar diameter as follows:
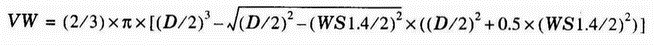
(8) where D is the diameter of the test ball (D = 6.00 mm).
- The relative wear (RW) was calculated as the ratio of the volume of ball wear obtained for the solution of fatty acid additive in the base fluid (VWfa) and the volume of ball wear for the base fluid (VWbf):
RW = (VWfa/VWfb) x 100% (9) - The force transducer attached to the lower specimen housing measured the friction force throughout a test. An A/D converter captured the friction force signal from the force transducer and the centre-line average value of the signal was calculated. That value was divided by the applied load to give the friction coefficient. The limiting value of the friction coefficient is not defined by CEC F-06-A-96.
- The electrical contact resistance (a semi-quantitative indicator of the formation of a separating film in the contact) was monitored throughout a test. The contact resistance circuit applied a 15 mV potential across the specimen contact and a balance resistor in series. The contact and the balance resistor thus formed a potential divider circuit. The series resistance was set by the control software and was set to 10 W by default. The potential drop across the contact was thus a measure of the contact resistance, as compared to the balance resistor. A low or zero film reading meant that the potential dropped across the contact, and hence the contact resistance was very low, i.e., there was a significant metal-to-metal contact taking place between the test specimens. This is usually associated with a high friction force and with high wear. A high film reading meant that the metal surfaces were apart. The limiting value of the film is not defined in CEC F-06-A-96.
Table 7. Tribological properties of tested diesel fuel components (HFR2 apparatus) Diesel fuel component Tribological wear Film % Corrected wear-scar
diameter, µmRelative ball wear Average value Standard deviation Average value Standard deviation HON component 691 6 100.0 21 4 HK component 703 3 100.0 13 2 Pin-on-disc test outputs Wear-scar diameter was measured in both the X and Y directions using a photomicroscope to an accuracy of ±1 µm, and the MWSD was evaluated. The influence of the absolute humidity was not taken into account, and the corrected wear-scar diameter was not determined. VW was calculated from the MWSD as follows:

(10) where D is the diameter of the test ball (D = 3.18 mm).
- The relative wear (RW) was defined in the same way as for the HFR2 test outputs.
RESULTS AND DISCUSSION
Test results
Influence of fatty acids on the trihological properties of low-sulphur diesel fuel components The tribological properties of two low-sulphur diesel fuel components were compared. The results of tests carried out using the HFR2 apparatus show that the HK component, containing a lower amount of sulphur, has slightly worse antiwear behaviour than the HON component (Table 7).- Tribological tests of 100, 500, and 1000 ppm stearic acid solutions in both components were performed using the HFR2 apparatus to determine the influence of small fatty acid additive amounts on the tribological properties of the components. The largest difference in wear value for these solvents appears at a concentration of 500 ppm. Furthermore, a 500 ppm solution of stearic acid has better antiwear characteristics in the HK component (Table 8).
Table 8. Effect of stearic acid on tribological properties of HON component and HK component (HFR2 apparatus) Stearic acid concentration, ppm Tribological wear Film % Corrected wear-scar
diameter, µmRelative ball wear Average value Standard deviation Average value Standard deviation HON component 100 493 3 25.8 62 5 500 409 2 12.2 87 2 1000 319 8 4.5 90 3 HK component 100 470 15 19.7 44 4 500 336 3 5.1 81 1 1000 331 7 4.8 89 1 - The HON component was chosen as a base fluid for preparing C6-C16 acid solutions in the concentration range 50-1000 ppm. Tests carried out for those solutions using the pin-on-disc tester show that the wear does not decrease with increased solution concentration for each acid tested, and the wear does not decrease with increased molecular weight of the acid for each concentration (Table 9).
- The test results obtained from the HFR2 apparatus for 500 ppm solutions of C6-C22 acids (Table 10 ) confirm the untypical relationship between wear and the number of carbon atoms in the fatty acid molecule (molecular weight) (Figure 1). Additionally, it is found (Figure 2) that there is a correlation between wear and film thickness for capric, lauric, palmitic, stearic, and behenic acids (C10-C22 fatty acids). However, for caproic acid, in spite of a thin film, the wear-scar diameter is relatively small.
Influence of fatty acids on tribological properties of separated fractions The HON component was studied in detail and separated into fractions (fraction I-X) by vacuum distillation. These fractions were tested using pin-on-disc and
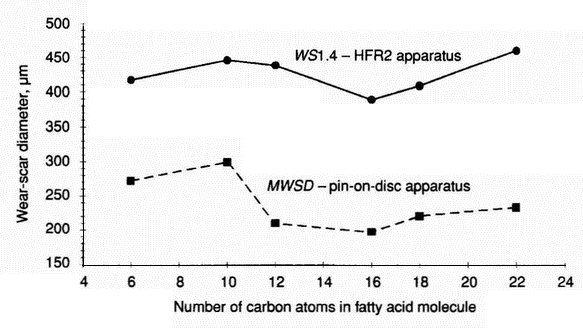
Figure 1: Tribological wear in systems lubricated with 500 ppm solutions of fatty acids in the HON component under different test conditions 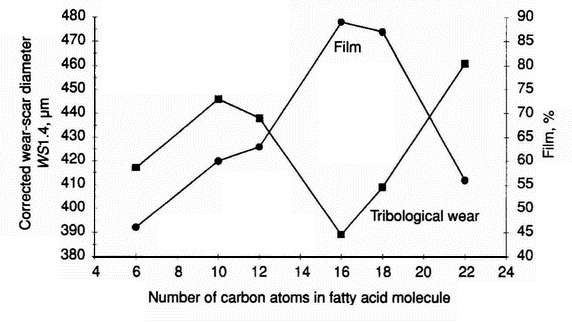
Figure 2: Tribological wear and film thickness in systems lubricated with 500 ppm solutions of C6-C18 fatty acids in the HON component (HFR2 apparatus) Table 11. Tribological wear in systems lubricated with distillation fractions of HON component (pin-on-disc apparatus) Fraction Tribological wear Wear-scar diameter, µm Relative ball wear % Average value Standard deviation I 522 33 144.2 II 609 37 267.9 III 582 36 224.4 IV 619 72 286.3 V 534 64 158.0 VI 594 46 243.7 VII 567 52 201.7 VIII 547 71 174.2 IX 527 55 150.4 X 237 28 6.1 Table 12 Tribological wear in systems lubricated with distillation fractions of HON component (HFR2 apparatus) Fraction Tribological wear Corrected wear-scar diameter, µm Relative ball wear % Average value Standard deviation I 757 14 144.0 II 780 24 162.6 III 770 30 154.1 IV 745 9 135.3 V 743 16 133.6 VI 723 22 119.7 VII 699 25 104.8 VIII 691 14 99.8 IX 659 16 82.3 X 533 15 35.3 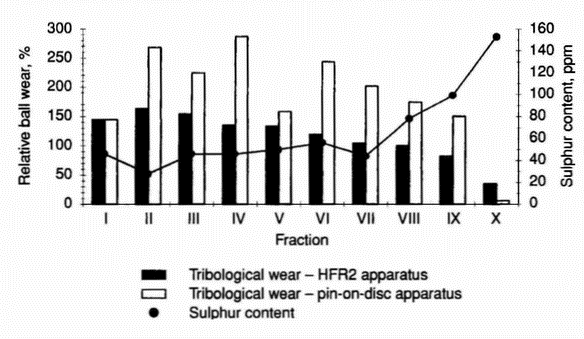
Figure 3: Influence of sulphur content on tribological wear in systems lubricated with distillation fractions of the HON component under different test conditions 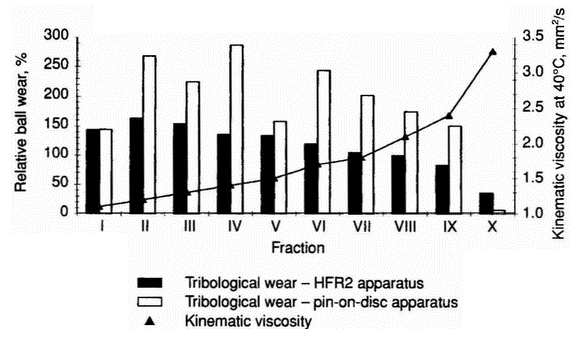
Figure 4: Influence of viscosity on tribological wear in systems lubricated with distillation fractions of the HON component under different test conditions 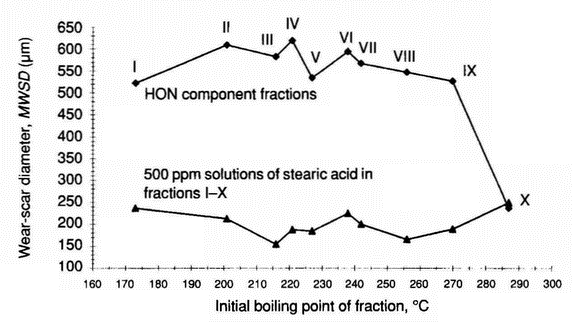
Figure 5: Tribological wear in systems lubricated with distillation fractions of the HON component and 500 ppm solutions of stearic acid in these fractions (pin-on-disc apparatus) HFR2 apparatus. On the basis of the pin-on-disc results, clear evidence was found that only the last fraction (fraction X) gives lower wear than the HON component (Table 11). However, the HFR2 test results show that fraction IX also has antiwear properties (Table 12).
- In order to study the factors influencing the tribological properties of fractions, the sulphur content and viscosity of fractions I-X were determined (Table 2). The relative ball wear obtained from tests carried out using the HFR2 apparatus decreases with increasing viscosity and correlates with sulphur content for all the fractions except fraction VII (Figures 3 and 4). The pin-on-disc test results indicate that for lighter fractions wear does not depend on sulphur content and viscosity; the difference in the wear values for fractions I-VI may be caused by the tribological properties of particular groups of hydrocarbons in individual fractions under these test conditions (Figures 3 and 4).
- Figure 5 shows wear in the systems lubricated with fractions I-X, and wear in the systems lubricated with 500 ppm solutions of stearic acid in these fractions plotted against the initial boiling point of the fractions. The addition of 500 ppm of stearic acid into fractions I-IX produces marked wear reduction but does not influence the tribological properties of fraction X (Table 13).
Influence of fatty acids on the tribological properties of different hydrocarbon base fluids Figure 6 (see p. 312) shows the relative change in wear for n-hexadecane, 1-methylnaphthalene, and their mixture containing palmitic acid
Table 13 Tribological wear in systems lubricated with 500 ppm stearic acid solutions in distillation fractions of HON component (pin-on-disc apparatus) Fraction Tribological wear Wear-scar diameter, µm Relative ball wear % Average value Standard deviation I 236 67 4.1 II 212 64 1.5 III 155 15 0.5 IV 188 36 0.8 V 184 32 1.4 VI 225 35 2.0 VII 200 47 1.5 VIII 166 17 0.8 IX 189 13 1.7 X 249 21 122.4
*The ratio of the volume of wear of the ball obtained for a fraction containing 500 ppm stearic acid additive and the volume of wear of the ball obtained for the fraction, multiplied by 100%.at different concentrations. In considering the influence of palmitic acid concentration on both 1-methylnaphthalene and its 1:1 ratio by volume mixture with n-hexadecane, three effects can be seen:
- a 100 ppm concentration of palmitic acid reduces the ball wear for both lubricants very significantly
- for the mixture of aromatic and paraffin hydrocarbons at a 750 ppm concentration of palmitic acid, a maximum value of wear appears, as in the case of n-hexadecane containing the same amount of acid (note that in Figure 6, this latter value is multiplied by 10 -1)
- no wear maximum appeared for 1-methylnaphthalene.
- The effect of viscosity on the tribological behaviour of fatty acids was studied using two polyalphaolefin base oils. Figure 7 shows that an
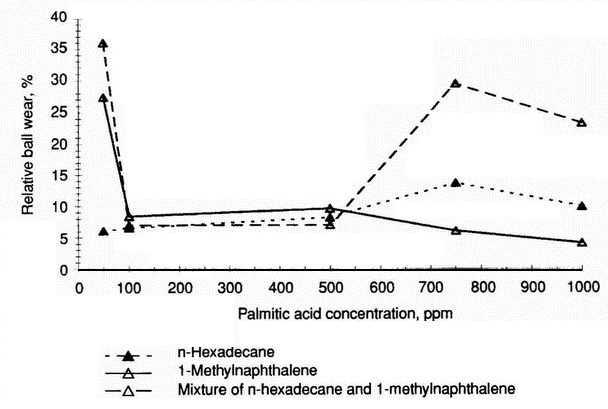
Figure 6: Relative ball wear in systems lubricated with 50-1000 ppm solutions of palmitic acid in n-hexadecane, 1-methylnaphthalene, and equivolume mixture of n-hexadecane and 1-methylnaphthalene (pin-on-disc apparatus) increase in the viscosity of the base oils decreases the wear reduction at all palmitic acid concentrations in the range 100-1000 ppm. Figure 7 demonstrates also that a 100 ppm addition of acid to synthetic base fluids PAO 4 and PAO 6 reduces the wear significantly, and at this acid concentration a minimum wear value appears.
- Interestingly, for high-viscosity base fluids with 100 ppm addition of acid, a clear maximum wear value was found (Table 14). Additionally, a 50 ppm concentration of palmitic acid in these base fluids also increased the wear. These trends are true for the saturated hydrocarbons (paraffin and naphthenic) separated from mineral base oil SN 400 and for the mineral base oil SN 400.
Discussion
Oxygen-containing impurities (e.g., naphthenic acids, phenols) are usually present in diesel fuels. Over the past few years a range of hydrogenation treatments of varying severity and hydrocracking processes have become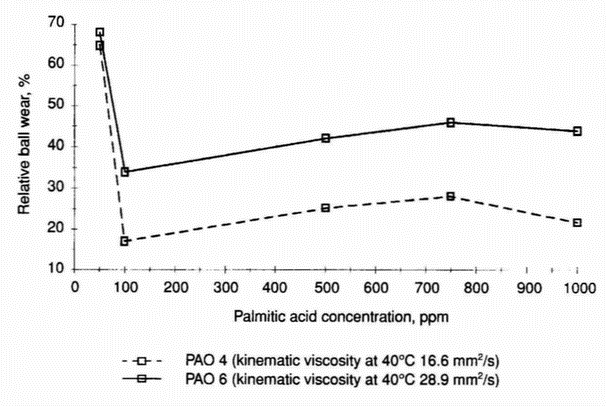
Figure 7: Relative ball wear in systems lubricated with 50-1000 ppm solutions of palmitic acid PAO 4 and PAO 6 (pin-on-disc apparatus) common. These are especially effective at reducing the concentration of polar compounds and are potentially responsible for a large decrease in fuel lubricity.
- Wei and Spikes [4] considered that the significant wear reduction was produced by oxygen compounds with phenolic-type or carboxylic acid groups and occurred at low concentrations (Table 15). Our studies confirm that fatty acids added to low-sulphur diesel fuel components in a concentration range of 50-1000 ppm produced excellent antiwear properties (Tables 8-10).
- Fatty acids of higher molecular weight, starting from lauric acid (C12), appear to be superior friction-reducing agents. A wide variety of early systematic studies [11, 13, 14] indicate that friction is reduced by the existence of a thin layer of a fatty acid material different from that of the contacting surfaces and which adheres strongly to the solid surface. Depending on the substrate surface, physical adsorption or chemisorption or both play an important part in friction-reducing film formation. At this point a question arises: what is the relation between friction and wear? According to Bowden and Tabor,13 although friction shows a strong correlation with wear, there is no simple general relation between the two. The fact that low friction does not necessarily mean low wear
Table 14 Wear data for model and real base fluids containing palmitic acid in the concentration range 0-1000 ppm (pin-on-disc apparatus) Base fluid Palmitic acid concentration, ppm Wear-scar diameter, µm Relative ball wear % Average value Standard deviation 1 -Methylnaphthalene 0 385 13 100.0 50 279 22 27.4 100 208 37 8.4 500 215 16 9.6 750 192 7 6.1 1000 174 9 4.2 Mixture of 1-methylnaphthalene and n-hexadecane 0 346 21 100.0 50 268 47 36.0 100 178 11 7.0 500 178 7 7.0 750 255 42 29.5 1000 241 43 23.4 PA04 0 281 35 100.0 50 252 37 64.9 100 181 9 17.0 500 199 41 25.2 750 205 59 28.1 1000 192 56 21.7 PA06 0 213 16 100.0 50 194 39 68.1 100 163 21 34.0 500 172 22 42.0 750 175 8 45.9 1000 174 7 43.9 Saturated hydrocarbon mixture from SN 400 0 212 23 100.0 50 232 17 145.2 100 238 14 159.3 500 205 16 88.3 750 190 26 65.4 1000 156 10 29.6 Mineral base oil SN 400 0 222 18 100.0 50 264 10 202.3 100 270 18 219.0 500 215 13 88.7 750 225 14 107.0 1000 167 15 32.4 Table 15 Influence of oxygen-containing compounds on tribological properties of hydrotreated diesel fuel [4] Fluid Oxygen from added compound, ppm Wear-scar diameter, µm Hydrotreated diesel fuel 0 350 Solution of 8-hydroxyquinoline in hydrotreated diesel fuel 10 320 Solution of 8-hydroxyquinoline in hydrotreated diesel fuel 100 180 Solution of 1,4-hydroquinoline in hydrotreated diesel fuel 100 220 Solution of caproic acid in hydrotreated diesel fuel 100 230 Solution of palmitic acid in hydrotreated diesel fuel 1 330 Solution of palmitic acid in hydrotreated diesel fuel 10 220 Solution of palmitic acid in hydrotreated diesel fuel 100 180 Solution of palmitic acid in hydrotreated diesel fuel 300 170 was emphasised over fifty years ago [19]. However, this fact is often overlooked and it has also contributed somehow to the lack of systematic and detailed studies on the influence of fatty acids on the wear of tribological mating elements. It has been shown, however, that although there is no simple, universally applicable relation between friction and wear, for specific systems, where effects such as ploughing and corrosion are negligible, a relation can be established [20]. It has been shown (Figure 2) that there is a correlation between wear and film thickness for capric, lauric, palmitic, stearic, and behenic acids (C10—C22) fatty acids). For caproic acid, in spite of a thinner film, the corrected wear-scar diameter is relatively small. For all fatty acids tested, the film thickness is greater than for neat diesel fuel components. Figure 8 shows wear and friction coefficient against number of carbon atoms and it can be seen that there is no correlation between wear and friction coefficient for C6—C22 fatty acids.
- In our opinion, two points seem to be of importance:
- a kind of molecular interaction exists between the fatty acid additive and the chemistry of the base fluids
- the viscosity of the base fluids seems to be a factor in the boundary lubrication process.
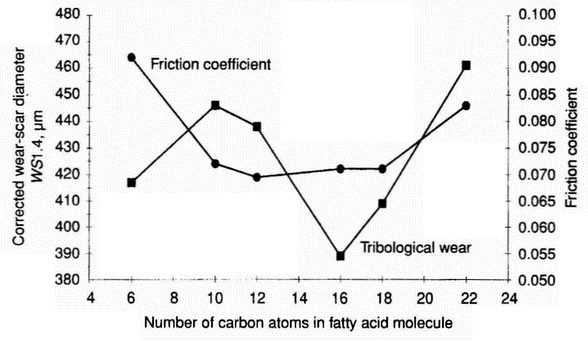
Figure 8: Tribological wear and friction coefficient in systems lubricated with 500 ppm solutions of C6-C22 fatty acids in the HON component (HFR2 apparatus) - Our previous research [21], in which n-hexadecane was used as the base fluid, indicated a new phenomenon concerning a very specific prowear and antiwear behaviour of fatty acids in the 50-1000 ppm concentration range. For this specific behaviour of fatty acids, two kinds of effects could be distinguished.
- a clear corrosive effect at a concentration of 750 ppm of myristic acid (C14), palmitic acid (C16), and stearic acid (C18)
- a distinct antiwear effect at a concentration of 500 ppm of caproic acid (C6).
- Results concerning 1-methylnaphthalene and its mixture with n-hexadecane (1:1 by volume) confirm that at a 100 ppm concentration of palmitic acid, a significant wear reduction of the steel-on-steel system can be achieved. For the mixture, the low-wear region extends to 500 ppm. At a 750 ppm concentration a maximum wear value appears (Figure 6); however, the maximum is located in the low-wear region (relative wear around 30%). In the case of 1-methylnaphthalene the increasing acid concentration slightly decreases the wear. Figure 7 demonstrates that for the synthetic base fluids, a clear minimum wear value exists at a 100 ppm concentration of palmitic acid; however, at a 750 ppm concentration of the acid, only a slight maximum is observed. In comparison with the wear data for n-hexadecane-based lubricants, both PAO 4- and PAO 6-based lubricants have these maxima at a relative wear reduction below 50%. Surprisingly, it was found that the addition of 50 and 100 ppm of palmitic acid to SN 400-saturated hydrocarbons increases wear by around 50% (Table 14). The same is true for the mineral base oil SN 400. However, in this case the corrosive effect is even larger; the wear is increased by a factor of two. When the concentration of palmitic acid was increased to 1000 ppm, the antiwear behaviour of both oils was improved (relative wear around 30%). The viscosity can influence the wear behaviour of fatty acids used at the lowest concentrations. For polyalphaolefin base oils, an increase in viscosity decreases wear reduction at all palmitic acid concentrations of 100-1000 ppm (Figure 7).
- All the above-discussed findings are challenging from the viewpoint of their interpretation as well as from their relation to the lubricating oil formulation. The results obtained evidently confirm the complexity of the boundary lubrication approach.
CONCLUSIONS
The results of the influence of 50-1000 ppm C6-C22 fatty acid solutions in diesel fuel components, their fractions, and other real and model solvents obtained from tribological tests carried out using a pin-on-disc tester and HFR2 apparatus have been described and discussed.- One of the most interesting conclusions is that fatty acids added to low-sulphur diesel fuel components in the concentration range 50-1000 ppm produce excellent antiwear properties.
- Interestingly, tribological wear does not decrease with increased concentration of any fatty acid in a diesel fuel component. It was also found that the fatty acid chain length was not a controlling factor in the wear process for either the pin-on-disc apparatus or the HFR2 apparatus.
- Palmitic acid solutions in real and model solvents show that the antiwear behaviour of fatty acids is the result of complex interactions between molecules of the acid and particular types of hydrocarbons. This has been confirmed by results obtained for palmitic acid solutions ofn-hexadecane, 1-methylnaphthalene, and mixtures of these hydrocarbons.
- The antiwear behaviour of fatty acids under boundary lubrication conditions is influenced by the rate of acid molecule diffusion to the friction surface. This conclusion is drawn from results of experiments obtained for systems lubricated with palmitic acid solutions in synthetic oils (polyalphaolefins) of various viscosities. It has also been found that in some cases the structure of the solvent molecule has a predominant influence on wear.
References
- Spikes, H.A., and Wei, D.-P., 'Fuel lubricity - fundamentals and review', Proc. 1st International Colloquium on Fuels, Technische Akademie Esslingen, Germany, 1997, pp. 249-58.
- Spikes, H.A., and Cameron, A., 'A comparison of adsorption and boundary lubricant failure', Proc. Roy. Soc. London Ser. A, 336 (1974) 407-19.
- Onion, G., and Suppeah, J.K., 'The generation and properties of boundary films formed by diesel fuel at a steel/steel conjunction', Trib. Int., 17 (1984) 277-87.
- Wei, D., and Spikes, H.A., 'The lubricity ofdiesel fuels'. Wear, 111 (1986) 217-35.
- Nikanjam, M.. and Burk, E., 'Diesel fuel additive study', SAE paper 942014, 1994.
- Wang, J.C., and Cusamo, C.M., 'Predicting lubricity of low sulfur diesel fuels', SAE paper 952564, 1995.
- Lacey, P.I., and Westbrook, S.R., 'Diesel fuel lubricity', SAE paper 950248, 1995.
- Mitchell, K.J., 'Diesel fuel lubricity - a survey of 1994/95 Canadian winter diesel fuels', SAE paper 961181, 1996.
- Wei, D.-P, and Spikes, H.A., 'Fuel lubricity - fundamentals and review'. Fuels International, 1, 1 (2000) 45-65.
- Hardy, W.B., Collected Works, Cambridge University Press, Cambridge, UK, 1936.
- Dorrison, A., and Ludema, K.C., Mechanics and Chemistry in Lubrication, Tribology Series 9. Elsevier, Amsterdam, 1985.
- Campbell, W.E., 'Boundary lubrication', in Boundary Lubrication - An Appraisal of World Literature, ed. F.F. Ling, E.E. Klaus, and R.S. Fein, ASME, New York, 1969, pp. 87-117.
- Bowden, P.P., and Tabor, D., Friction and Lubrication of Solids, Clarendon Press, Oxford, 1950.
- Jahanmir, S., 'Chain length effect in boundary lubrication'. Wear, 102 (1985) 331-49.
- Bellzer, M., and Jahanmir, S., 'An adsorption model for friction in boundary lubrication', ASLE Trans. 29, 3 (1986) 423-30.
- Wachal, A., 'The influence of chemical structure of additives on antiseizure and antiwear properties of oils', Tribologia, 8, 5-6 (1977) 161-3 (in Polish).
- Kajdas, C., Luczkiewicz, J., Ozimina, D., and Wawak-Pardyka, E., 'The influence of concentration of alcohols and acids on tribological properties of paraffin oil', Tribologia, 11, 6 (1980) 22 (in Polish).
- Wills, J.G., Lubrication Fundamentals, Marcel Dekker, New York, 1980.
- Larsen, R.G., and Perry, G.L., 'Investigation of friction and wear under quasi-hydrodynamic conditions', Trans. ASME, 67 (1945) 45-50.
- Rabinowicz, E., 'The friction on boundary-lubricated surface', in Proc. 2nd Nat. Cong. on Applied Mechanics, 1954, pp. 595-9.
- Kajdas, C., Majzner, M., and Konopka, M., 'The effect of fatty acids at low concentrations in n-hexadecane on wear of steel in boundary lubrication', Tribologia, 153, 3 (1997) 221-37 (in Polish).
A revised version of a paper first presented at the 2nd International Colloquium on Fuels, Technische Akademie Esslingen, Germany.
- Additionally, tribological tests were carried out for distillation fractions obtained by fractional separation of the HON component (properties of fraction I-X are summarised in Table 2).